Particle Size Distribution and Its Measurement
The population of particles in a powder, as described by its particle size distribution (PSD), affects the properties of a powder and dispersions in many important ways. For example, in industrial applications, it determines the setting time of cement, the hiding power of pigments, the activity of catalysts, the taste of food, and the sintering shrinkage of metallurgical compositions. The relevance to the pharmaceutical industry is that the stability, aesthetics, rate of absorption and total bioavailability, etc. are all strongly affected by the size of the particles that comprise the powder or dispersion. In addition, regulatory agencies are becoming increasingly aware of the importance of the PSD and requiring developers to have greater control and understanding of this aspect of their drug products.
What is Particle Size?
Surprisingly, it is not a question that can be easily answered. This is because in the real world, depending upon the material properties, conditions of manufacture and storage, three distinct species (Figures 1 and 2) will always exist in any powder, wet or dry1 -the existence of which results from a variety of different binding mechanisms.
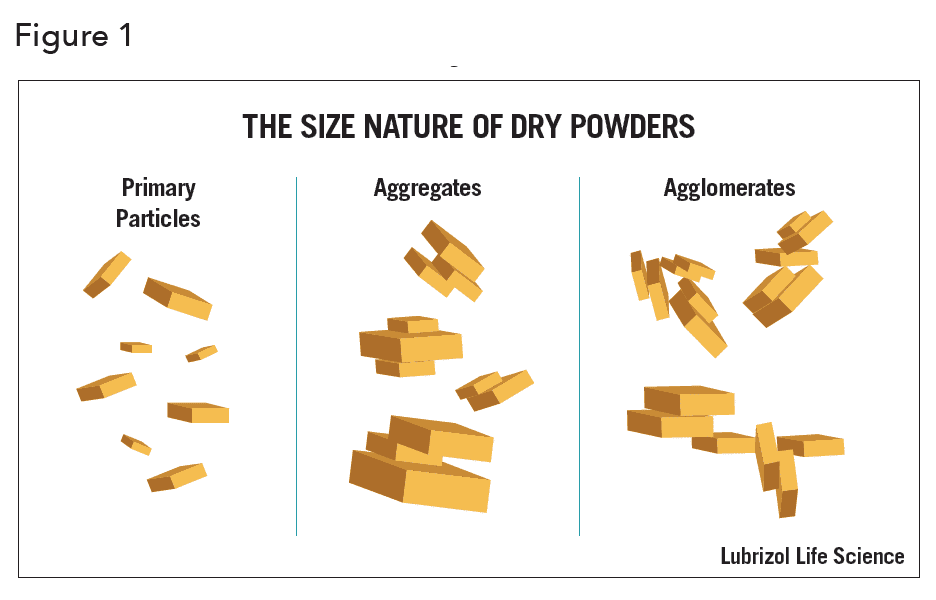
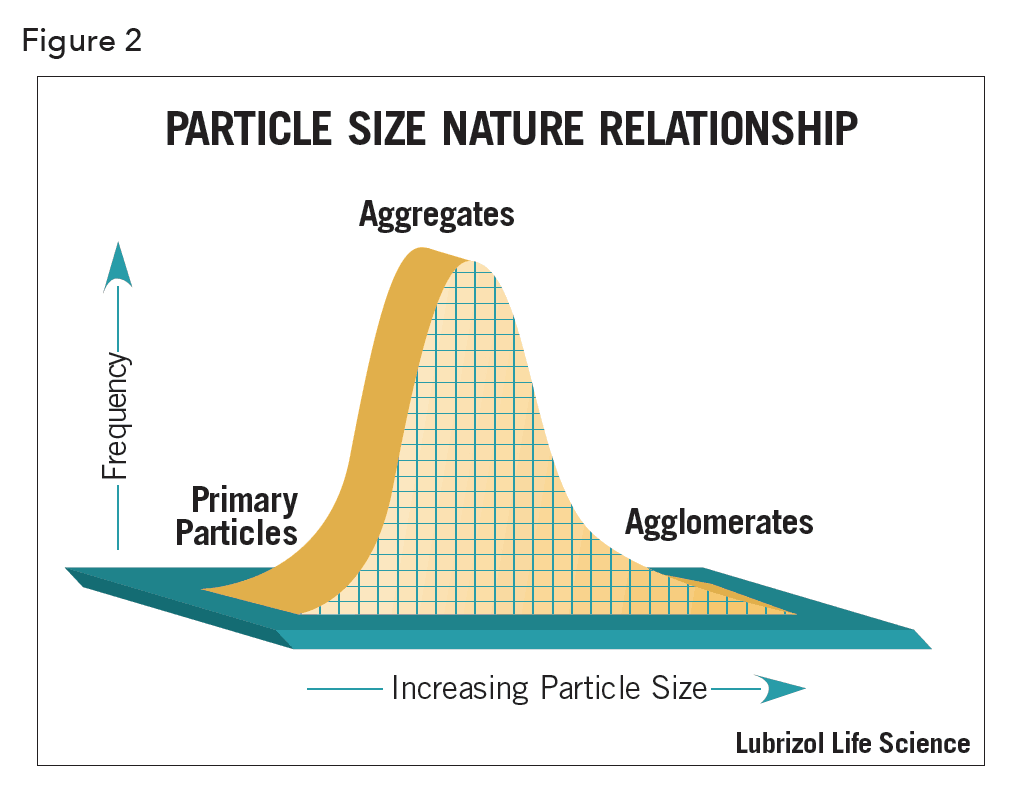
The three species are:
- Primary particles. These are inorganic or organic structures held together by atomic or molecular bonding. They are the “fundamental” particles. Primary particles cannot be separated into smaller particles except by the application of ultrahigh energy. In any sample they are usually present at only a fraction of a percent.
- Aggregates. These comprise two or more primary particles tightly bound together by rigid chemical bonding resulting from sintering or cementation. Aggregation is the coalescence of particles by processes other than heat/pressure, i.e., precipitation of ionic salts onto surfaces during manufacture. Aggregates are typically formed when powders are heated, compressed, or dried from a suspension. They have a large interfacial area of contact between each particle and the force necessary to rupture these bonds is considerable. Aggregates constitute, for all practical purposes, the largest single fraction of any particle size distribution (PSD) that one can hope to achieve in a formulation.
- Agglomerates. These are collections of aggregates, loosely held together at point-to-point contact by weak electromagnetic forces, van der Waals forces, mechanical friction, and interlocking. Agglomerates are formed when fine particles are handled, shaken, rolled or stored undisturbed in a single position. They can readily be broken apart with proper dispersion techniques.
During the formulation process, while it is critical to obtain an initial suspension in which the particles are narrowly distributed, it is equally important that the PSD remains the same at the end of any formulating process. The end result of a correct dispersing process is to effectively narrow the PSD: to reduce the agglomerates to aggregates. Starting with a “pre-dispersed” system cannot guarantee that one will end up with the same distribution. Any such dispersion will have a predetermined set of dispersants, stabilizers and excipients that may or may not be compatible with the rest of the formula’s ingredients. The PSD in the final drug product is what will determine the performance of the PSD-dependent parameters and thus, it is the PSD in the final drug product that must be controlled and measured.
This is no small task and it has to be recognized from the outset that there is no exact, or accurate, measure of “a particle size”. There are, instead, measures of PSD at a given time under given conditions. All powders (and suspensions thereof) exist as a PSD; in other words, there exists a range of sizes. However, the PSD can be quantified and a single value ascribed. If understood, such a number can be useful, especially in quality control (QC) efforts. Such a single value is, however, of little use in the development process since it can hide any number of complicating events. The PSD needs to be understood in detail during development and, once characterized, can be described by a single number that is sensitive enough to indicate a change and trigger corrective action.
For QC, there are three single numbers that can be used: the mean (or average), the mode, and the median. The mean is the arithmetic mean of the area under the PSD “curve”. The modal value is that size where the majority of particles are located. The median is value at which 50% (by whatever is being reported, mass for instance) of the particles are greater and 50% smaller. For narrow Gaussian distributions these three values are often approximately the same. This is usually found to be the case with reference standards such as (spherical) polymer latexes. For the majority of suspensions of “real world” materials however, such as active pharmaceutical ingredients (APIs), the distribution is skewed and the three values can be vastly different. It is important to understand the PSD so that one can determine which of the three values correlates best with end-use performance.
Particle Size Measurement
Only in the case of a single sphere can the size of a particle be completely described by one number – its diameter. For other regular shapes it is usually necessary to specify more than one dimension; a cuboid is defined by length, width and height. For the vast majority of materials, particles are rarely spherical. For such irregular particles the assigned “size” will depend upon the method of measurement2. With commercial particle sizing instrumentation the most widely used descriptor is the “equivalent spherical diameter”, i.e., the diameter of a sphere of equivalent volume. Thus, a unit cube has the same volume as a sphere of diameter 1.24 units.
The proliferation of measuring techniques has arisen because of the wide range of sizes and size dependent properties that have to be measured; a twelve-inch ruler is not a satisfactory tool for measuring either mileage or thousandths of an inch. Likewise, an instrument designed to measure particles several hundred microns in diameter is unlikely to be of help in determining the real PSD of nanometer-sized powders. Further complication arises because often the size range of the particles in any one system may be too wide to be measured with any one measuring device.
Therefore, when deciding which technique to use, the analyst must first consider the purpose of the analysis. What is generally required is not the size of the particles, per se, but the value of some property of the particles that is size-dependent. More often than not in drug development, a given behavior, such as bioavailability, is correlated with a particular particle size. For ongoing production then, the ability to detect a change in PSD is far more important than an absolute particle size determination. When investigating PSD versus property X, it is critical to clearly establish the goal and not get lost chasing what can be irrelevant levels of accuracy at the expense of repeatability and sensitivity. In such circumstances it is important, whenever possible, to measure the desired property, rather than to measure the “size” by some other method and then deduce the required property. For example, instead of measuring the “size” of catalyst particles, it is the surface area that should be determined since this is the property that determines the reactivity of the catalyst. In short, the method should measure the appropriate property of the particles, with accuracy sufficient for the particular application.
Currently, the most widely used technique for PSD determination is that of laser light scattering. There are two variations: dynamic light scattering (DLS)3 and Fraunhofer diffraction (FD)4. The choice is dictated by the size range under investigation. DLS works for sizes from a few nanometers up to about one micron (1,000 nm) and FD works from about one micron up to several millimeters. DLS has become the de facto method of particle size analysis of APIs but is increasingly becoming inadequate with the ever-growing number of nanoparticulate-based drug products. Both methods are non-imaging and are called “ensemble averaging” methods. This means that the two relevant pieces of information needed to describe the PSD (namely the actual size and the amount of material at that size) need to be deconvoluted from a single measurement of the amount of light scattered. This involves application of extremely complex theory and equally complex deconvolution algorithms5. Thus, both measurement variations are intrinsically low resolution: typically, the best that can be achieved is to differentiate between two class sizes6. A major problem arises when the PSD extends across the “one-micron divide”, because then two different theories/algorithms need to be applied but cannot be combined. Attempts to do so result in artifacts in the PSD such that commercial instruments smooth the data resulting in even less resolution. Being based on light scattering, the fundamental value obtained is an “intensity weighted” number (DI). Two major problems arise in transforming the DI value into a Dv (“volume-weighted” number), which is a necessary requirement when measuring the PSD of APIs since drugs are administered by volume or mass. The first is that, in addition to the assumption of sphericity, both DLS and FD “weight” the intensity differently (d6 and d2, respectively). The second is that the transform necessitates application of scattering “corrections” – again both different. The result is that the Dv value (and, if further transforms are made to spherical-equivalent diameter, Ds, and number diameter, Dn) can be widely inaccurate. However, for QC purposes, the techniques are extremely fast and, for measurements on the same material, extremely reproducible.
Sample preparation
It is, undoubtedly, the case that much of the variation in particle sizing measurements is traceable to either incorrect sampling or sample preparation. Particle size analysis results are only applicable when any samples drawn are representative and the appropriate dispersion techniques are used. Sampling is an important subject in its own right2,7 and will be discussed separately.
When preparing suspensions of powders in liquids, there are many assumptions made by both novices and experienced formulators. Dispersion has been described as the “forgotten parameter”. While it is by no means a straightforward process, it is not an art. It is an exact scientific discipline based on established physicochemical principles which is controlled by a variety of factors, each of which needs to be considered separately and in the correct sequence8.
Thus, when comparing measurements of PSD it is critical that, one, the dispersion process used be the same (i.e., type of dispersion aid, mechanical treatment, etc.), and two, the measurement technique be the same. Otherwise measurement can become a meaningless exercise. Lastly, and perhaps most important, whatever the preparation technique is, it must remain sensitive to product changes that may alter those parameters that are determining safety and efficacy.
References
- G. Lawrence, “Dispersion of Powders in Liquids”, (Ed.), G.D. Parfitt, Third Edition, Applied Science Publishers, London (1981).
- Allen, “Particle Size Measurement”, Fourth Edition, Chapman and Hall, New York (1990).
- Pecora, “Dynamic Light Scattering: Applications of Photon Correlation Spectroscopy”, Plenum Press, New York (1985).
- Swithenbank, et al, “Prog. Astronaut. Aeronaut.”, 53 421 (1977).
- Kerker, “The Scattering of Light and other Electromagnetic Radiation”, Academic Press, New York (1969).
- B. Weiner in “Particle Size Distribution – Assessment and Characterization”, (Ed.), T. Provder, ACS Symposium Series No.332, American Chemical Society (1987).
- Gy, “Sampling of Particulate Material, Theory and Practice”, Elsevier, Amsterdam (1982).
- Fairhurst and M. Mitchnick, “Sunscreens: Development, Evaluation and Regulatory Aspects”, (Eds.), N.J. Lowe, N.A. Shaath, and M.A. Pathak, Volume 2, Marcel Dekker (2000).
